FAris Livingstone
8-15-14
Flame Test LAb
Purpose
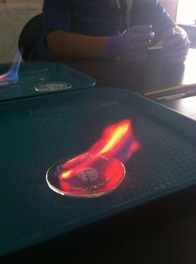
In this lab we were experimenting with fire, chemical compounds, and methanol. We were looking at the color of fire after pouring methanol on different chemical compounds and lighting them. We also have to make observations before and after the fire to see how our chemical compounds physical appearance changed, if they were rocky, powdery, fluffy, white, black or gray.
Procedure
For this lab we used different chemical compounds mixed with methanol to make fire in different colors. These are the chemical compounds we used: Methanol (control), Borax, Copper Sulfate, Magnesium Sulfate, Strontium Chloride and an Unknown compound. Before we could light stuff on fire we had to assign roles and be safe. There were two people in each group, one note taker and one experimenter and we had to make sure everyone had goggles, aprons, and gloves (if experimenting).
Pre-Lab Questions
1. What color of light is the lowest in energy?
Red has the biggest wavelength which makes it the color of the lowest energy.
2.What color of light is the highest in energy?
Violet has the smallest wavelength which makes it the light with the highest energy.
3. What color of light is the highest frequency?
Violet light has the highest frequency.
4. What color of light is the lowest frequency?
Red light has the lowest frequency.
5. How are electrons “excited”?
They get excited after absorbing energy.
6. What does it mean when the electrons are “excited”?
When they are excited it means that they are attracted to something or each other.
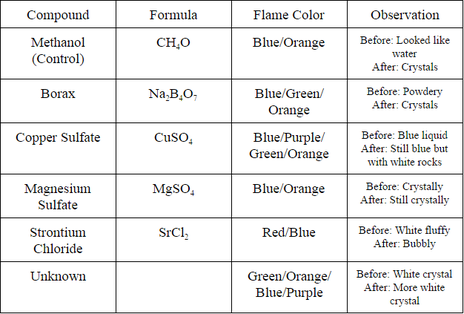
Data Tables and Observations
In this lab we were experimenting with fire, chemical compounds, and methanol. We were looking at the color of fire after pouring methanol on different chemical compounds and lighting them. We also have to make observations before and after the fire to see how our chemical compounds physical appearance changed, if they were rocky, powdery, fluffy, white, black or gray.
Procedure
For this lab we used different chemical compounds mixed with methanol to make fire in different colors. These are the chemical compounds we used: Methanol (control), Borax, Copper Sulfate, Magnesium Sulfate, Strontium Chloride and an Unknown compound. Before we could light stuff on fire we had to assign roles and be safe. There were two people in each group, one note taker and one experimenter and we had to make sure everyone had goggles, aprons, and gloves (if experimenting).
Pre-Lab Questions
1. What color of light is the lowest in energy?
Red has the biggest wavelength which makes it the color of the lowest energy.
2.What color of light is the highest in energy?
Violet has the smallest wavelength which makes it the light with the highest energy.
3. What color of light is the highest frequency?
Violet light has the highest frequency.
4. What color of light is the lowest frequency?
Red light has the lowest frequency.
5. How are electrons “excited”?
They get excited after absorbing energy.
6. What does it mean when the electrons are “excited”?
When they are excited it means that they are attracted to something or each other.
Data Tables and Observations
Post-Lab Questions1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
- I think the chemicals have to be heated so that certain particles from the chemical compounds are separated from the compound and burn in the flame. These particles mix with the heat of the flame to produce colors.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
- Ionic bonds are usually created when a non-metal and metal are combined. There is an electrostatic attraction between oppositely charged ions.
3. What is the definition of a salt in chemistry?
- A salt in chemistry is an ionic compound that is created due to the neutralization of an acid and a base. (princeton.edu)
4. What is fire?
- Fire is a burning of something. Particles chemically combine with oxygen to create heat, smoke and fire.
5. What are the chemicals involved in lighting a match?
- When lighting a math you are rubbing potassium chlorate against phosphorus which chemically mix to ignite something.
6. What is a chemical reaction?
- A chemical reaction is when the molecular or ionic structure of a chemical substance is rearranged.
7. What natural cycles does wildfire have an impact on?
- Wildfires can affect our water cycles by contaminating our water with debris and ash which makes our water unhealthy to use. They can also affect the carbon cycle because of the trees.
8. List at least three impacts of the wildfire in San Diego.
- The water and air was contaminated near our house so we had to wear oxygen masks and get water bottles from the store.
- It was very hot for a number of days because the temperature just kept on rising as the fire continued to blaze.
- It cleared away the dead trees so that new ones could take it's place and grow to start their new life.
Conclusion
In this lab I some cool new stuff about chemicals and chemical compounds, also about the chemical reaction between the fire and compound. I am still really curious about how a color is produced through the compound. Also what would happen if you combined all the compounds together, what color would the flame be. These are all some crazy question I still have left in my mind and can't wait to get answered.
- I think the chemicals have to be heated so that certain particles from the chemical compounds are separated from the compound and burn in the flame. These particles mix with the heat of the flame to produce colors.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
- Ionic bonds are usually created when a non-metal and metal are combined. There is an electrostatic attraction between oppositely charged ions.
3. What is the definition of a salt in chemistry?
- A salt in chemistry is an ionic compound that is created due to the neutralization of an acid and a base. (princeton.edu)
4. What is fire?
- Fire is a burning of something. Particles chemically combine with oxygen to create heat, smoke and fire.
5. What are the chemicals involved in lighting a match?
- When lighting a math you are rubbing potassium chlorate against phosphorus which chemically mix to ignite something.
6. What is a chemical reaction?
- A chemical reaction is when the molecular or ionic structure of a chemical substance is rearranged.
7. What natural cycles does wildfire have an impact on?
- Wildfires can affect our water cycles by contaminating our water with debris and ash which makes our water unhealthy to use. They can also affect the carbon cycle because of the trees.
8. List at least three impacts of the wildfire in San Diego.
- The water and air was contaminated near our house so we had to wear oxygen masks and get water bottles from the store.
- It was very hot for a number of days because the temperature just kept on rising as the fire continued to blaze.
- It cleared away the dead trees so that new ones could take it's place and grow to start their new life.
Conclusion
In this lab I some cool new stuff about chemicals and chemical compounds, also about the chemical reaction between the fire and compound. I am still really curious about how a color is produced through the compound. Also what would happen if you combined all the compounds together, what color would the flame be. These are all some crazy question I still have left in my mind and can't wait to get answered.